A clinical trial led by the Kirby Institute at UNSW Sydney has found that point-of-care testing for hepatitis B DNA is as effective as traditional laboratory testing, paving the way for faster diagnosis and treatment in hard-to-reach communities, as well as with the broader population.
Hepatitis SA has long championed the power of point-of-care testing (POCT). It means that a simple fingerstick blood test can quickly and accurately determine somebody’s viral hepatitis status, even in decentralised or remote settings. Over the last two years, Hepatitis SA has been actively working with SA Health and other services rolling out hepatitis C point of care testing in community settings accelerating progress towards hepatitis C elimination.
Point-of-care testing for hepatitis B, on the other hand had been much slower in becoming available. The first hepatitis B POCT clinic in South Australia was in November 2024, as part of a sub-study by the National Australian Hepatitis C Point-of-Care Testing Program but there has been little movement since.
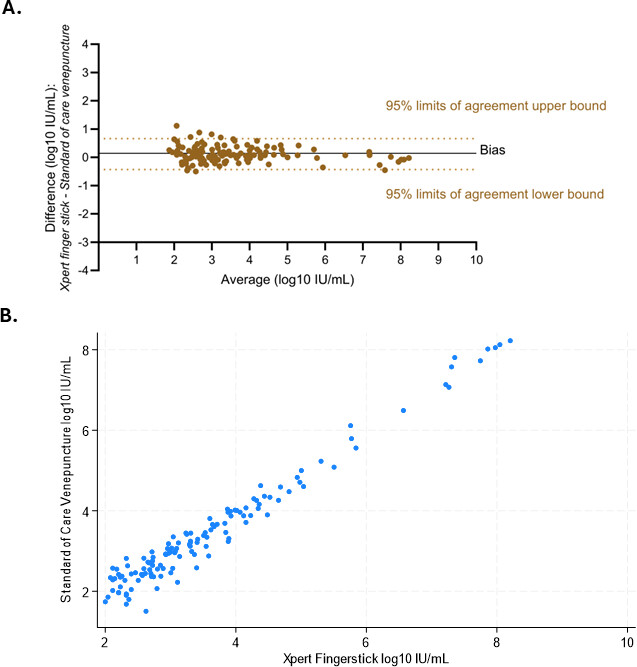
Results from the new Kirby Institute research – published in the Journal of Clinical Microbiology – has shown that hepatitis B point-of-care testing is as effective as the current standard-of-care.
“The results of our trial found that the fingerstick point-of-care test is highly accurate, closely matching the accuracy of traditional tests,” explained Professor Gail Matthews, who led the research at the Kirby Institute. “This is a very important finding because it has the potential to expand access to testing and treatment globally, and especially in resource limited settings or remote areas, where current testing access is poor.”
The results of this research also dovetail nicely with the recent release of new hepatitis B care guidelines issued by the World Health Organization, which recommend POCT assays and reflex HBV testing.
While most high-income countries have strong vaccination programs and reasonable access to care, the majority of people with chronic hepatitis B live in low- and middle-income countries, where access to testing and treatment is limited. A similar disparity exists between communities in high-income countries. In Australia for instance, hepatitis B DNA testing is more difficult to access for people living in remote areas.
“Not everyone who has hep B needs treatment,” explained Associate Professor Behzad Hajarizadeh, one of the researchers who wrote the paper. “People with higher levels of the virus are more likely to benefit from treatment, so DNA tests are required to determine the levels of virus in the system. DNA testing is also used once a patient starts treatment, to help understand if the treatment is working.”
Currently, hepatitis B DNA testing, for both diagnosis and monitoring, usually consists of collecting a venous blood sample to be processed in centralised laboratories, meaning some patients may need to travel long distances to take the test, and then often wait days or weeks for results.
This delay, and the multiple clinic visits involved, can hinder timely treatment and care, or mean that some patients put off or even never attend appointments. Fingerstick POCT changes all of that.
“Our research demonstrates that point-of-care testing for hepatitis B DNA using finger stick blood is, indeed, highly accurate and effective. Given the technology is already in use for a range of other infectious diseases globally, our evidence paves the way for integrating infectious disease care significantly enhancing access to hepatitis B testing, monitoring and treatment, no matter where someone lives,” explained the Kirby Institute’s Associate Professor Tanya Applegate.
Most recent World Health Organization figures (2022) estimate that there are 254 million people living with chronic hepatitis B infection worldwide, yet only 14 per cent were diagnosed and just eight per cent were receiving treatment, representing a major global health challenge. It is currently estimated that no country is on track to meet the WHO target of elimination of hepatitis B as a public health threat by 2030.
As part of a push to increase testing and treatment, most recent WHO guidelines include a new recommendation supporting the use of hepatitis B point-of-care DNA fingerstick tests globally. Data from this study supports that recommendation.

“Access to testing is a major barrier to progress on hepatitis B elimination,” says Associate Professor Thomas Tu from Hepatitis B Voices. “We are hopeful that this research will support the roll-out of point-of-care testing for hepatitis B, enhancing access and ultimately, improving health and saving lives.”
Hepatitis SA Outreach and Support Coordinator , Lisa Carter, agrees. She has witnessed how the GeneXpert testing has transformed access to care. “It is especially great for those who have difficulty accessing veins for blood tests,” she explains. “It is such a relief for them not to have to go through that to know their hepatitis status.”
Last updated 3 June 2026
More from:
Enjoyed this article? Subscribe to be notified whenever we publish new stories.
Subscribe for Updates









